Open Evidence is taking part in a research project led by Pfizer and the University of Oslo to ensure safe use of medicines
The initiative aims to provide European citizens with the information they need to actively manage their personal health and adhere to treatmentsOpen Evidence, a UOC spin-off, is one of the 40 organizations involved in this international project
On Monday 16th November, the Innovative Medicines Initiative (IMI) launched a 5-year worldwide research project - Gravitate-Health, that aims for improved access and understanding of health information, for safe use of medicines, risk minimization and improved quality of life.
40 leading partners from civil society, academia and industry in 15 countries have joined forces to develop novel, patient-focussed healthcare information tools that will offer citizens timely access to trustworthy, relevant information about medicines, based on the assertion that the engagement of patients in their own health relies on actionable, understandable, relevant, reliable and evidence-based information that meets their specific needs, health context, and literacy level. This unique public – private strategic partnership is co-led by the University of Oslo (public partner coordinator) and Pfizer (industry lead).
Safe use of medication and adherence to treatment are significant public health challenges where understanding, access to accurate information, capabilities to comprehend, and trust play major roles. OECD estimates that around 200,000 premature deaths annually in Europe relate to poor medication adherence, which may cost as much as 125 billion € each year in avoidable hospitalization, emergency care and adult outpatient visits[1]. Empirical studies of information flows in health care show lack of interoperability and broken chains of activity which complicate information sharing. Medication information can be fragmented, unavailable or not used, so introducing risks of which the user may be unaware, for example risk of errors in use that can lead to adverse events, disability, and even death.
Medicinal product information is subject to strict regulation and scientific validation procedures; it plays a pivotal role in assisting health care professionals when prescribing and dispensing the medicine, and informs patients and consumers about its safe use. Currently, patients primarily receive this information in the form of a paper leaflet provided with their medicine. This presents a range of practical challenges to access and understanding: the content of the leaflets may not be readily understandable or indeed relevant to the needs of many patients, is typically presented in small font sizes and may not reflect the most up-to-date information due to long production timescales for product packaging. These problems are likely to be aggravated by aging, multimorbidity and chronic diseases, with many patients needing to refer to multiple paper leaflets for different medicines.
Making regulator-approved medicinal product information from a trusted authorized source digitally available to patients and healthcare professionals is fundamental to support the digital transformation of healthcare across the EU, which will in turn offer significant public health benefits. It brings new opportunities to address shortcomings in the current patient information paradigm by allowing for ease of access to trustworthy content, speed of update, accessible formats, the ability to better personalize content to the needs of the individual and also to link with other health information systems, e.g. electronic health records.
To advance on this journey and address current challenges, Gravitate-Health will develop and test an integrated, digital, user-centric health information solution seeking to demonstrate tangible improvements in availability and understanding of health information from a set of trusted sources, starting with regulator-approved medicinal product information and the Electronic Health Record International Patient Summary (EHR-IPS). The main outputs will be an open source digital platform supporting G-Lens functionally, and a White Paper with recommendations on realistic strategies to strengthen access, understanding and future use of digital services like electronic product information (ePI) as a tool for risk minimization.
By developing electronic tools to improve access of patients and healthcare professionals to information on medicines Gravitate-Health aligns its goals with the relevant recommendations from the European Commission (COM(2017) 135 final) and the European Medicines Agency’s subsequent action plan to improve the product information for the EU (EMA/680018/2017).
The Action seeks to demonstrate that such improvement translates to higher levels of adherence to treatment, safer use of medication (Pharmacovigilance), and better health outcomes and quality of life, and to develop new and deeper insights into how use of available health information can be optimized to act as effective risk minimization measures. Patient voice and human touch are at the heart of Gravitate-Health. Broad patient and stakeholder engagement underpin the planned work and will drive the technology development through the life of the project.
“Our mission is to equip and empower patients with digital health information and tools that make them confident, active, and responsive in their patient journey, specifically encouraging safe use of medicines for better health outcomes and quality of life,” said Prof Anne Moen, coordinator of Gravitate-Health and Professor in the Institute for health and society, University of Oslo.
Gravitate-Health Project Lead, Dr Giovanna Ferrari of Pfizer, added: “This collaboration between academia, industry and stakeholder associations promises strong and unique results and an exciting opportunity to deliver new digital information approaches demonstrated to offer wide benefit to patients.”
Supported by a grant from IMI
The project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 945334. This joint undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and the European Federation of Pharmaceutical Industries and Associations [EFPIA]. The total budget is 18.5 M€ for a project duration of 60 months.


Project facts
- Project acronym: Gravitate-Health
- Start date: 01 November 2020
- Duration: 60 months
- Budget: €18,5 Mio
- Coordination: University of Oslo, Prof Anne Moen
- Project Lead: Pfizer Ltd, Dr. Giovanna Ferrari
For further information: https://www.oecd-ilibrary.org/docserver/health_glance_eur-2018-en.pdf
Gravitate-Health partners at a glance
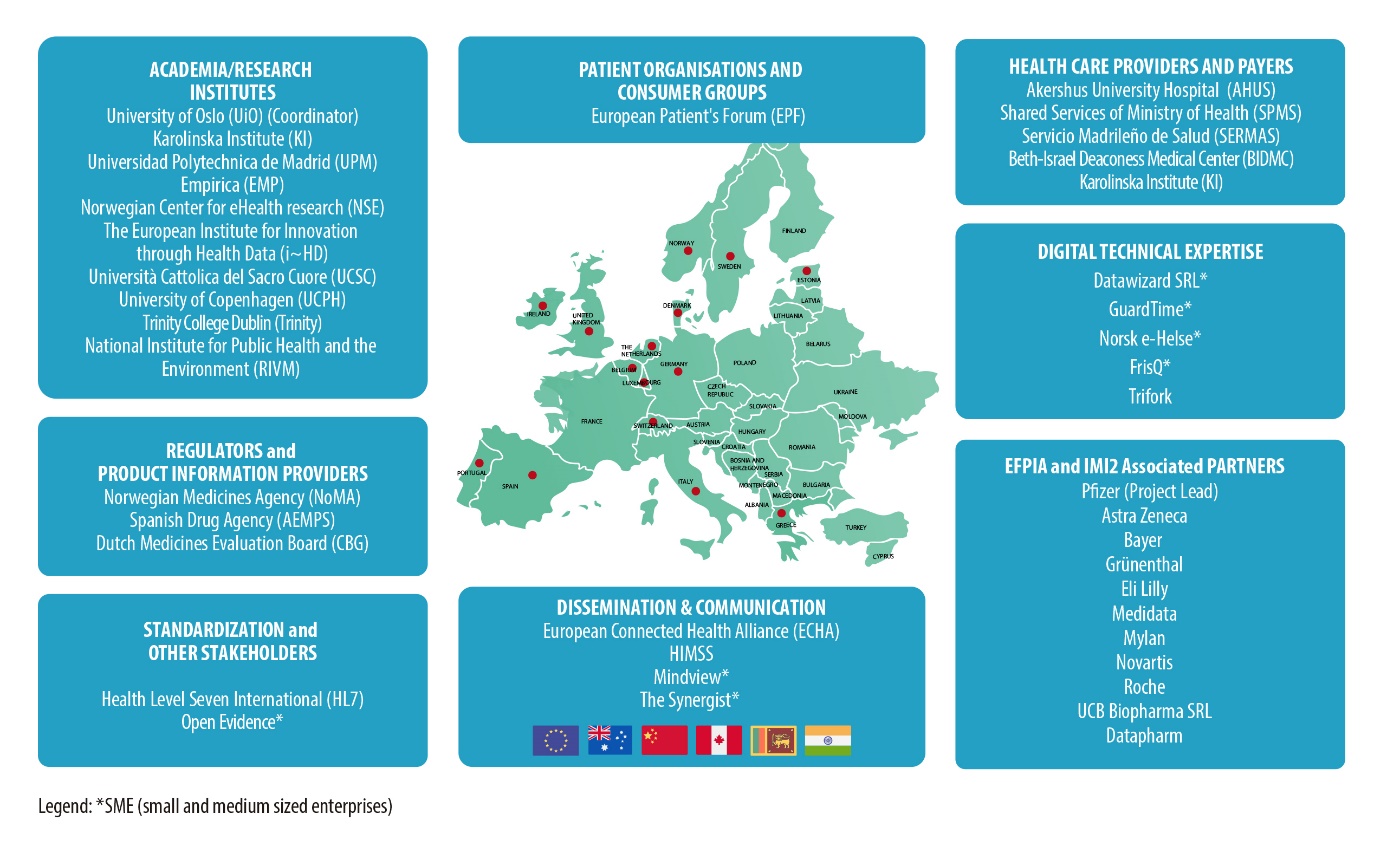
- Norway
- Universitet i Oslo (Coordinator)
- Norwegian Centre for eHealth Research
- Akershus Universitetssykehus
- Norsk e-Helse AS
- Statens Legemiddelverk
- United Kingdom
- Pfizer Limited (Project Lead)
- Eli Lilly and Company Limited
- MDSOL Europe Ltd
- Datapharm Limited
- Sweden
- Karolinska Institutet
- FrisQ Holding AB
- AstraZenica AB
- Denmark
- Københavns Universitet
- Trifork Public A/S
- Belgium
- HL7 Europe
- Forum European des Patients
- The European Institute for Innovation through Health Data
- The Synergist
- UCB Biopharma SRl
- Italy
- Datawizard SRL
- Università Cattolica del Sacro Cuore
- Spain
- Universidad Politécnica de Madrid
- Open Evidence
- Servicio Madrileño de Salud
- Agencia Española de Medicamentos y Productos Sanitarios
- Germany
- empirica Gesellschaft für Kommunikations-und Technologieforschung mbH
- HIMSS Europe GmBH
- Bayer Aktiengesellschaft
- Grünenthal GmBH
- Ireland
- Trinity College Dublin
- European Connected Health Alliance
- Mylan IRE Healthcare Limited
- The Netherlands
- Rijksinstituut voor Volksgezondheid en Milieu Agentschap Collegee ter Beoordeling van Geneesmiddelen
- Greece
- MINDVIEW Business Consulting and Research limited
- Portugal
- Serviços Partilhados do Ministério da Saúde, E.P.E.
- Estonia
- Guardtime OÜ
- Switzerland
- Novartis Pharma AG
- F. Hoffmann – La Roche AG
- USA
- Division of Clinical Informatics at Beth Israel Deaconess Medical Center
About IMI
The Innovative Medicines Initiative (IMI) is Europe’s largest public-private initiative aiming to improve health by speeding up the development of, and patient access to, innovative medicines, particularly in areas where there is an unmet medical or social need. IMI facilitates collaboration between the key players involved in healthcare research, including universities, the pharmaceutical and other industries, small and medium-sized enterprises (SMEs), patient organisations, and medicines regulators. It is a partnership between the European Union (represented by the European Commission) and the European pharmaceutical industry (represented by EFPIA, the European Federation of Pharmaceutical Industries and Associations). For further information: www.imi.europa.eu. UOC R&I
About R&I
The UOC's research and innovation (R&I) are helping 21st-century global societies to overcome pressing challenges by studying the interactions between ICT and human activity, with a specific focus on e-learning and e-health. Over 400 researchers and 50 research groups work among the University's seven faculties and two research centres: the Internet Interdisciplinary Institute (IN3) and the eHealth Center (eHC).
The United Nations' 2030 Agenda for Sustainable Development and open knowledge serve as strategic pillars for the UOC's teaching, research and innovation. More information:research.uoc.edu. #UOC25years
DISCLAIMER: The present press release reflects the authors’ view and neither IMI nor the European Union, or EFPIA, re responsible for any use that may be made of the information contained herein.
Press contact
-
Editorial department
